When inventors are granted a patent, they may believe they have taken all of the crucial steps to protect themselves and their inventions. However, this is not necessarily the case. In order to provide maximum protection for their patent, paten owners must provide notice of patent protection in order to maximize the potential damages the owner can seek if the patent is infringed. Notice is provided by properly marking their patented products, packaging, or labels.
Governed by 35 U.S. Code § 287, the statute states, “Patentees, and persons making, offering for sale, or selling within the United States any patented article for or under them, or importing any patented article into the United States, may give notice to the public.” There are two different methods that allow patent owners and licensees to comply with the law: (1) traditional patent marking; and (2) virtual patent marking.
Traditional Patent Marking
Traditional patent marking requires that the patent number is clearly identified on the product or its packaging if marking the product is not feasible (e.g. the product is too small). This method of patent marking may present an issue when patent portfolios grow, and patents expire over time. When changes occur, the patent marks on the products will need to be updated, which can be quite burdensome, due to the effect of the product’s manufacturing process. However, if a company’s product is covered by only one patent, it may be more feasible to use this traditional patent marking method since the marking would not need to change in the future.
In order to comply with the statute, patent owners must use the term “Patent” or “Pat.” followed by the patent number. For example, the mark might read, “Pat. 9999999.” If a company’s patent expires and it continues selling products with the expired patent listed on them, it risks facing legal fees due to false patent marking.
One way to avoid false patent marking is by marking items on their labels rather than on the products themselves. That way, when a patent expires, companies can reproduce updated labels rather than having to waste products with false marks and manufacture new ones with updated patent numbers.
Additionally, if a list of patents covers a family of products, patent owners can mark the products by stating, “Covered by one or more of Patents: US9999999, US8888888, US7777777.” Every listed patent does not have to cover every member of the product family. Below is an example of a of toner cartridge by Brother which utilized this method.

Virtual Patent Marking
In 2013, the America Invents Act enacted virtual patent marking, offering a presumably more practical way to patent mark products in today’s society. Patent holders can place a webpage address on the packaging or label that leads to a site of associated patent numbers when putting the patent number on the product itself is not feasible. This method requires that the product or its packaging clearly identifies a web address that would indicate where one could find the patents for the product. For example, the product can be marked, “Pat.: https://www.camelbak.com/en/legal/patents.”
On the webpage, patent owners should make the patented article easy to find by utilizing pictures, product identification numbers, product numbers or any other tactics so that visitors can find the information quickly. For example, one way to display the information is by creating a table with products defined by their names in one column and a correlating patent number beside it as demonstrated on Keurig’s site below.
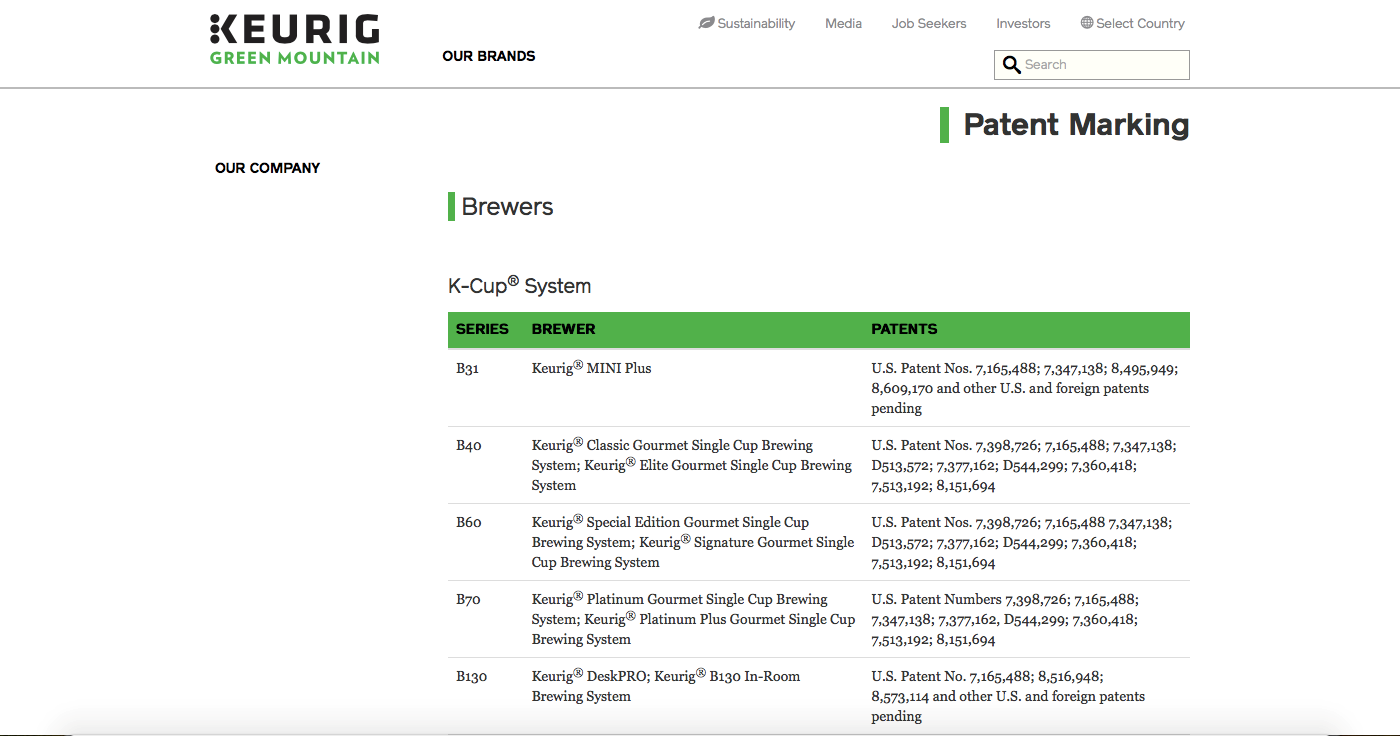
(See http://www.keuriggreenmountain.com/en/Patents.aspx) As mandated by the statute, those who decide to utilize the virtual method must make the webpage accessible to the public and cannot require a click-through agreement, nor a fee or payment, and must make the process as straightforward as possible.
Similar to the traditional marking method, patent holders can virtual mark a family of products by stating, “Covered by one or more of Patents:” followed by a web address where the patents can be found.
The advantage of virtual patent marking is that regardless of what happens with the patent portfolio in the future, the products’ packaging will remain the same.
Patent License Marking Requirements
Not only does patent marking apply to patent owners, it also applies to those who license the patented products. If a licensee fails to mark the products, a patent owner may be unable to collect past damages for infringement prior to actual notice of the infringement. Because of this, it is essential that patent owners ensure its licensees are complying with the patent marking statute by establishing the need to patent mark the items in the licensing agreement. This is an essential term to place in a licensing agreement.
Recently, in Rembrandt Wireless v. Samsung Elecs. (Fed. Cir. 2017), the court found that Samsung infringed upon two of Rembrandt’s patents, awarding Rembrandt $15.7 million in damages. However, Samsung appealed, arguing that Rembrandt’s patent was licensed to a third party, and it did not require the licensee to mark the products. The Federal Circuit remanded the case, deciding that damages would only be awarded to Rembrandt for post-notice damages, and that pre-notice damages would now be excluded.
Overall, patent owners should make reasonable efforts to guarantee compliance with the statutory marking requirements. Companies should work with a transactional attorney when drafting their license agreement, as the language and terms of the agreement could dictate who, how, and if the company can defend a licensed patent.
False Patent Marking & its Consequences
Companies should be aware that wrongly associating a patent number on a product will result in consequences governed by 35 U.S. Code § 292. Those falsely marking a product with a wrong patent number or marking “patent pending” when no patent application has been made will risk being fined up to $500 for each offense.
Marking a product with “patent pending” does not provide any benefits, such as the right to sue others for damages, but rather it simply notifies the public that you have filed a patent application with the U.S. Patent & Trademark Office. By notifying the public of the patent application, a competitor may be deterred from copying the idea.
Benefits to Patent Marking
By properly marking their products, whether utilizing the traditional or virtual method, patent owners give actual notice of the existence of their patents to the world. Regardless of whether an infringer possessed actual knowledge of the patent, patent marking allows patent owners the ability to collect damages from an infringer. Therefore, infringers cannot claim ignorance about the patent and will be liable to the patent owner for damages. Following the statute allows patent owners to immediately seek damages or redress after an infringement is discovered.
If the patent owner has not yet marked its products and the patent has been allegedly infringed upon, the owner will not be able to recover damages unless the infringer was notified and continued to infringe. (An action for infringement would constitute putting the infringer on notice or sending a letter, for example.) Damages would be recoverable, but only after the notice of infringement. Damages incurred before the lawsuit was filed would not be recoverable.
When patent owners fail to mark their products correctly, they have failed to put the public on actual notice and have given up their rights to collect damages. The law assumes that because no indication was provided on the patented product, the alleged infringer could not have known about the patent. Therefore, infringers will not be liable for patent infringement until they have actual knowledge of the patent. The law is structured this way in order to encourage patent owners to comply with the law so that upon infringement, patent owners will be eligible to collect damages.
Although placing patent numbers on brochures or websites, for example, may notify the public in general about your patent and may assist a patent owner in marketing its products, those types of measures will not satisfy the requirements to receive benefits after infringement under the statute. The patent mark must be on the product or its packaging.
As detailed above, complying with the patent marking statute, including taking reasonable measures to ensure that licensees are also complying, is essential to providing actual notice to the public and to secure the ability to seek infringement damages from not only the beginning of the lawsuit, but more importantly, damages from the time the infringement began. It is essential to seek legal counsel to determine the best way to mark your product.
 It’s a frustration many can relate to. You’re on the go with your smartphone, juggling business and personal calls and texts, when you suddenly realize you’re low on power. No worries. Just dip into a friendly café with your charger and power up while you are getting coffee’d up. So you reach into your bag for the charging cable …
It’s a frustration many can relate to. You’re on the go with your smartphone, juggling business and personal calls and texts, when you suddenly realize you’re low on power. No worries. Just dip into a friendly café with your charger and power up while you are getting coffee’d up. So you reach into your bag for the charging cable … How did you discover the NYSSTLC/Innovation Law Center and the services it provides businesses and entrepreneurs?
How did you discover the NYSSTLC/Innovation Law Center and the services it provides businesses and entrepreneurs?






